1. Metastasis-on-Chip
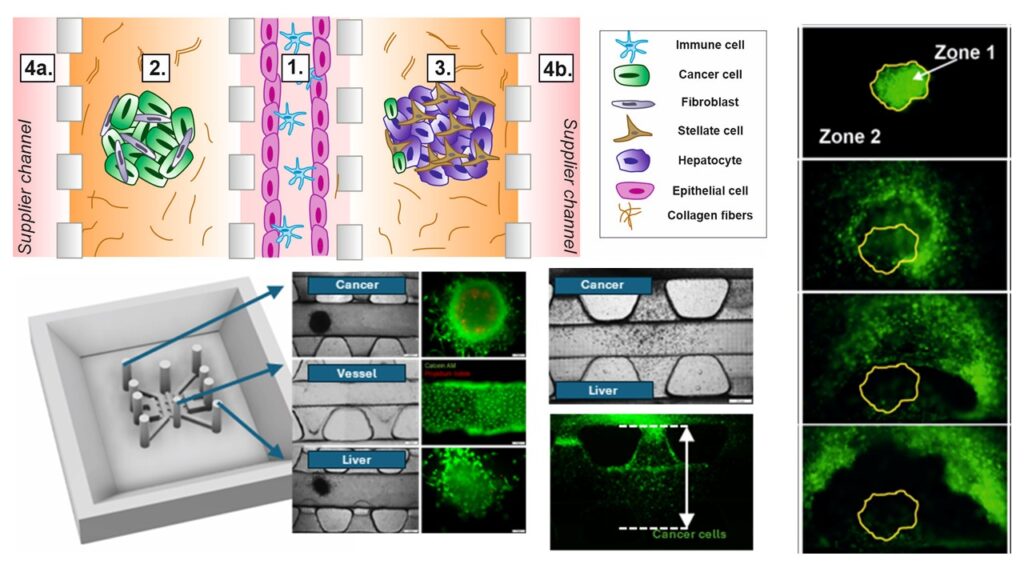
Cancer metastasis is the spread of cancerous cells through the circulatory system to distant organs. Existing in vitro models remain insufficient to faithfully reproduce the metastatic process. Multi-Organ-on-Chip (multi-OoC) platforms allow the integration of complex tissue models. Here, we propose a microplatform that recapitulates breast cancer (BC) migration to the liver, considering an endothelial barrier (EB) and immune cell interactions. Tissue micromodels were created using agarose multi-wells, loaded into the microplatform, and separated by different types of barriers: (i) collagen type I, (ii) cell culture medium, (iii) immune (Jurkat) cells, (iv) a microvessel, and (v) a microvessel perfused with Jurkat cells. Spatial arrangement of cells, their morphology, and viability were imaged using fluorescence microscopy over 10-day experiments. Quantitative data such as Feret Diameter, relative Raw Integrated Density (ID) and migration distance of tumor cells (GFP-MDA-MB-231) were evaluated. The concentrations of metastatic agents (interleukin-6 (IL-6), and interleukin-11 (IL-11)) were determined using ELISA. The potential of a microplatform in drug screening was preliminarily assessed with the use of Doxorubicin (Dox) over a 7-day experiment. Changes in Feret diameter and ID indicated a gradual disintegration of the BC micromodel. BC cells migrated toward the liver micromodel through a barrier formed in the central microchannel. An EB was impenetrable for GFP-MDA-MB-231, whereas Jurkat cells promoted the migration of BC cells. Dox induced transient inflammation and suppressed IL-11-dependent pro-metastatic signaling, consistent with its dual cytotoxic and immunomodulatory roles.
People involved in the project:
Publications:
2. Multi-Organ interaction in cancer treatment
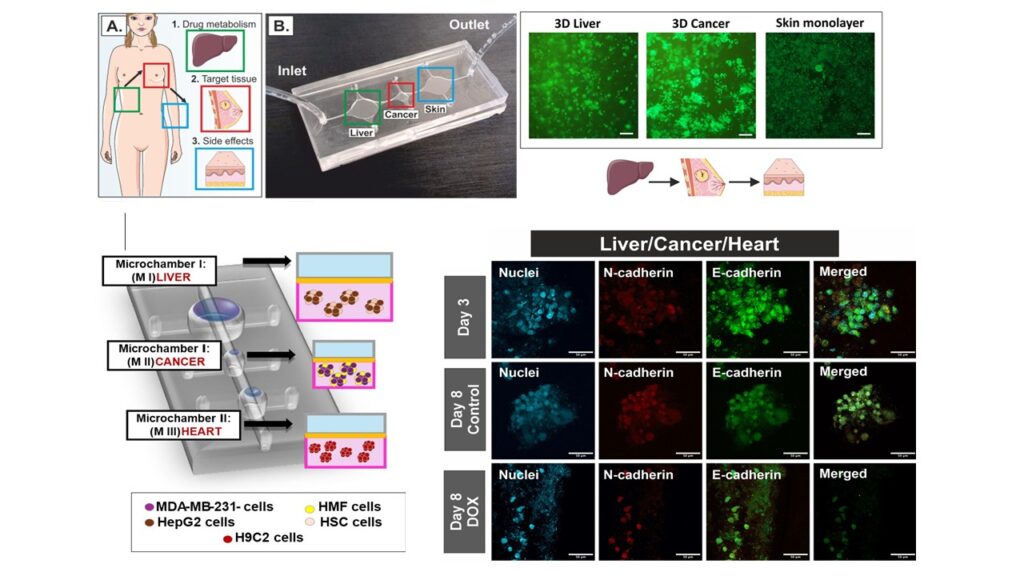
Cancer is one of the pathological conditions of the human body, which, due to its tissue diversity, is not fully understood. Currently used preclinical in vitro cell or animal models do not reflect the complexity and functional features of the human body, including its pathological conditions such as cancer. This fact is related to poor predictions of the effectiveness of newly developed drugs. Therefore, in our work, we focused on creating a tool that allows the reproduction of important morphological and biochemical features of the tumor in vivo, such as three-dimensional (3D) structure, heterogeneity, the presence of extracellular matrix (ECM), and the appropriate scale (volume to surface ratio). Moreover, the presented Multi-Organ-on-Chip (MOC) tool allows us to evaluate the effects of anticancer therapy, considering hepatic metabolism (liver model) and the assessment of its side effects on a selected organ (skin model or heart model). Our research shows that incorporating multiple organ models in one in vitro tool affects the viability and metabolic activity of the cells that constitute them. Moreover, we have shown how important it is to consider hepatic metabolism when evaluating the therapeutic effectiveness of selected anticancer drug.
People involved in the project:
Publications:
- Ulanowicz G., Gluchowska M., Zuchowska A. ; Standard in vitro co-culture conditions for studying inter-organ interactions between liver, breast cancer, and heart models; Chemico-Biological Interactions 2025, 419, 111659.
- Ulanowicz G., Galecka V., Romanczuk P., Kołodziejek D., Jastrzebska E., Brzozka Z., Zuchowska A. ; multi-Organ-on-Chip based on microfluidic approach to understanding organ interactions in cancer therapy; Advanced Materials Technologies, 2025, 0:e01748.
- Romanczuk P., Zajda J., Matczuk M., Zuchowska A. ; Multi-Organ-on-Chip approach to study the impact of inter-organ communication on the efficacy and side effects of cancer therapy; Chemico-Biological Interactions 2025, 413, 111460.