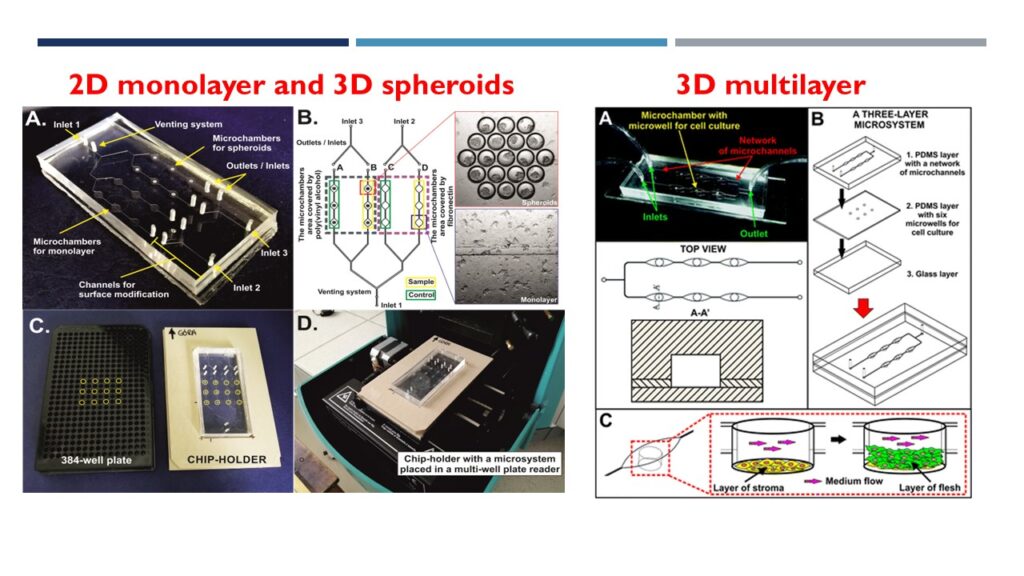
This research focuses on the development of advanced Tumor-on-Chip platforms that integrate microfluidic technology with both two-dimensional (2D) and three-dimensional (3D) cancer models to more accurately recapitulate the native tumor microenvironment. These systems enable precise control over fluid dynamics, nutrient and oxygen gradients, and cell–cell as well as cell–matrix interactions, thereby providing a more physiologically relevant alternative to conventional in vitro models.
Tumor-on-Chip approach represents a powerful and predictive tool for studying cancer biology and enhancing the preclinical evaluation of anticancer therapies, with strong potential for improving translational research and personalized medicine.
1. Drug testing
By employing both mono- and co-culture configurations, the Tumor-on-Chip platforms capture key aspects of tumor complexity, including cellular heterogeneity and microenvironment-driven responses. These models in our group have been widely applied to evaluate the efficacy of photodynamic therapy (PDT), incorporating advanced photosensitizers such as nanoencapsulated compounds and graphene-based nanocarrier systems. In addition, the platforms have been used to investigate the effects of conventional cytotoxic drugs, as well as combination therapies, enabling a more comprehensive assessment of treatment strategies.
People involved in the project:
- Elżbieta Jastrzębska
- Agnieszka Żuchowska
- Magdalena Flont
- Zbigniew Brzózka
Publications:
- Flont M., Mackiewicz Z., Białek M. Dybko A., Jastrzębska E.: A layered cancer-on-a-chip system for anticancer drug screening and disease modeling, Analyst, RSC Publications, 148, 21, 2023, 5486-5495, DOI:10.1039/d3an00959aFlont M.
- Flont M., Jastrzębska E.: A Multi-Layer Breast Cancer Model to Study the Synergistic Effect of Photochemotherapy, Micromachines, 14, 9, 2023, 1806, 1-17, DOI:10.3390/mi14091806
- Flont M., Jastrzębska E., Brzózka Z.: A multilayered cancer-on-a-chip model to analyze the effectiveness of new-generation photosensitizers, Analyst, 2020, vol. 145, no. 21, pp.6937-6947. DOI:10.1039/D0AN00911C
- Flont M., Jastrzębska E., Brzózka Z.: Synergistic effect of the combination therapy on ovarian cancer cells under microfluidic conditions, Analytica Chimica Acta, 2020, vol. 1100, pp.138- 148. DOI:10.1016/j.aca.2019.11.047
- Żuchowska A., Buta A., Dąbrowski B., Jastrzębska E., Żukowski K., Brzózka Z.: 3D and 2D cell models in a novel microfluidic tool for evaluation of highly chemically and microbiologically pure graphene oxide (GO) as an effective drug carrier, Sensors and Actuators B – Chemical, 2020, vol. 302, pp.127064_1-127064_10. DOI:10.1016/j.snb.2019.127064
- Zuchowska A., Kasprzak A., Dabrowski B., Poplawska M., Brzozka Z.; Nanoconjugates of graphene oxide derivatives andmeso-tetraphenylporphyrin: a new avenue for anticancer photodynamic therapies – Cell-on-a-Chip analysis; New Journal of Chemistry 2020, 44, 18770.
- Tokarska K., Bazylińska U., Jastrzębska E., Chudy M., Dybko A., Wilk K., Brzózka Z.: Selective cancer-killing ability of new efficient porphyrin-based nanophotosensitizer in Lab-on-a-chip system, Sensors and Actuators B – Chemical, 2019, vol. 282, pp.665-674. DOI:10.1016/j.snb.2018.11.115
- Chudy M., Tokarska K., Jastrzębska E., Bułka M., Drozdek S., Lamch Ł., Wilk K., Brzózka Z.: Lab-on-a-chip systems for photodynamic therapy investigations, Biosensors & Bioelectronics, 2018, vol. 101, pp.37-51. DOI:10.1016/j.bios.2017.10.013
- Żuchowska A., Marciniak K., Bazylińska U., Jastrzębska E., Wilk K., Brzózka Z.: Different action of nanoencapsulated meso-tetraphenylporphyrin in breast spheroid co-culture and mono-culture under microfluidic conditions, Sensors and Actuators B – Chemical, 2018, vol. 275, pp.69-77. DOI:10.1016/j.snb.2018.08.037
- Żuchowska A., Jastrzębska E., Chudy M., Dybko A., Brzózka Z.: 3D lung spheroid cultures for evaluation of photodynamic therapy (PDT) procedures in microfluidic Lab-on-a-Chip system, Analytica Chimica Acta, 2017, vol. 990, pp.110-120. DOI:10.1016/j.aca.2017.07.009